Most people assume that if a drug is approved and on the shelf, it’s been thoroughly tested for safety. But here’s the truth: drug interactions often don’t show up until millions of people are taking them. Clinical trials involve a few thousand volunteers over months - not years - and rarely include older adults, pregnant women, or people with multiple health conditions. That’s why some of the most dangerous drug combinations are only discovered after the drug is already in widespread use.
Why Clinical Trials Miss These Interactions
Pre-market trials are designed to prove a drug works, not to catch every possible side effect. A typical Phase 3 trial includes 1,000 to 5,000 people, lasts 6 to 12 months, and excludes those with liver or kidney disease, heart conditions, or those taking other medications. But real life isn’t like that. Most patients on statins also take blood pressure pills. Older adults often take five or more drugs. And many people drink grapefruit juice with their morning meds - something no trial would ever test for. The result? About 50-60% of common side effects get caught before approval. But 70-80% of serious, life-threatening reactions are only found after the drug hits the market. That’s not a flaw in the system - it’s a limitation of scale. You can’t simulate 10 million users in a lab.Three Types of Post-Market Drug Interactions
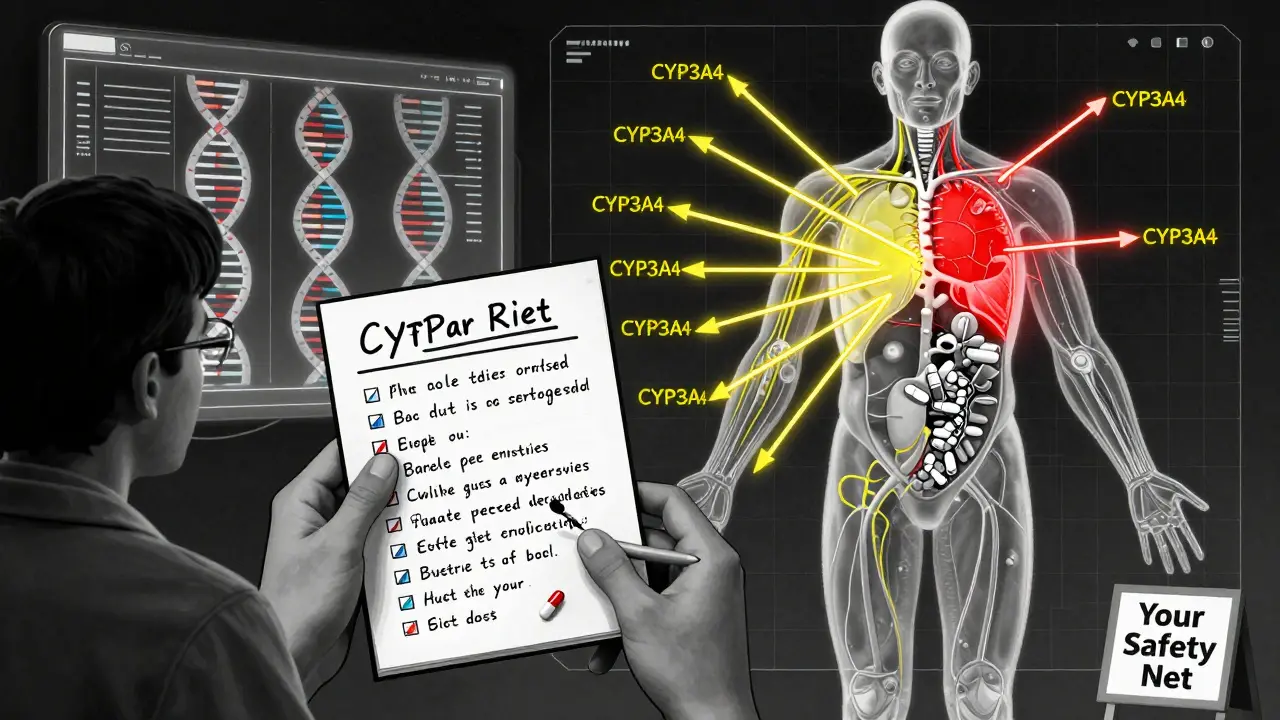
After a drug is approved, three main types of interactions start showing up:- Drug-drug interactions: One medication changes how another is absorbed, metabolized, or eliminated. For example, fluconazole (an antifungal) blocks the enzyme CYP3A4, which breaks down simvastatin. When taken together, simvastatin levels can spike 3 to 10 times higher - leading to rhabdomyolysis, a condition where muscle tissue breaks down and can cause kidney failure. This combo alone was linked to over 1,000 hospitalizations in the U.S. between 2015 and 2020.
- Drug-condition interactions: A health condition changes how a drug behaves. For instance, patients with chronic kidney disease who take apixaban (Eliquis) for atrial fibrillation may accumulate the drug in their system because their kidneys can’t clear it. This increases bleeding risk. Many patients weren’t warned because their kidney function wasn’t monitored during trials.
- Drug-food interactions: What you eat can make or break a drug. Grapefruit juice blocks CYP3A4 enzymes just like fluconazole. One study found it raised atorvastatin (Lipitor) levels by up to 15 times. That’s why some patients end up in the ER with muscle pain, dark urine, and kidney damage - all because their doctor never mentioned the grapefruit warning.
Real Cases That Changed Drug Safety Rules
Some interactions didn’t just cause harm - they led to drug withdrawals and black box warnings.- Terfenadine (Seldane): This once-popular allergy drug was pulled from the market in 1998 after it was found to cause fatal heart arrhythmias when taken with ketoconazole (an antifungal) or even grapefruit juice. The interaction was rare - but deadly.
- Benfluorex (Mediator): Used in France for weight loss, this drug was linked to heart valve damage in over 5 million patients over 30 years. It wasn’t withdrawn until 2009 - after hundreds of deaths.
- Pergolide (Permax): A Parkinson’s drug withdrawn in 2007 after being tied to heart valve disease in about 1 million patient-years of use.

How These Risks Are Caught
The FDA doesn’t just wait for complaints. Its FAERS (FDA Adverse Event Reporting System) collects over 1 million reports annually from doctors, pharmacists, and patients. But here’s the problem: experts estimate 90-95% of actual adverse events go unreported. That’s why the agency also uses real-world data. The Sentinel Initiative, launched in 2008, monitors over 300 million patient records from hospitals, insurers, and clinics. It uses algorithms to spot unusual spikes - like a sudden jump in rhabdomyolysis cases linked to a specific statin and antifungal combo. When a signal pops up, the FDA investigates. Sometimes, they update labels. Sometimes, they issue public warnings. Rarely, they pull the drug. In 2023, the FDA approved its first AI-powered pharmacovigilance platform that can analyze 10,000 reports a day with 92.7% accuracy. The European Medicines Agency now uses similar tools to cut signal detection time from 18 months to just 45 days.What This Means for You
If you take more than one prescription, you’re at risk. Even if your doctor didn’t mention an interaction, it might still exist.- Always ask your pharmacist: “Could this new drug interact with anything else I’m taking?”
- Check for grapefruit juice warnings on statins, blood pressure meds, and immunosuppressants.
- Don’t assume natural supplements are safe. St. John’s Wort, for example, can reduce the effectiveness of birth control, antidepressants, and blood thinners like apixaban. One FDA report in 2022 described a 78-year-old man who bled internally after mixing St. John’s Wort with Eliquis - a warning that wasn’t clear on the label.
- Use tools like GoodRx or Medscape’s drug interaction checker. One user on Reddit said, “The interaction warning prevented me from taking ciprofloxacin with my blood pressure meds - my pharmacist confirmed it could have caused dangerous QT prolongation.”
The Hidden Cost of Missed Interactions
Adverse drug events cost the U.S. healthcare system over $3.5 billion a year. About $1 billion of that comes from interactions that were never caught before approval. Hospital admissions from these errors account for 15-20% of all drug-related hospital stays. Pharmaceutical companies now spend billions on post-market surveillance. The global pharmacovigilance market grew from $5.8 billion in 2020 to $7.3 billion in 2022. Companies with over $1 billion in revenue have entire teams dedicated to monitoring safety signals. Smaller biotechs outsource this to firms like Parexel, which made $1.2 billion in pharmacovigilance revenue in 2022 alone.What’s Changing - and What’s Not
The FDA now requires post-approval studies for 45.7% of new drugs, and 22.3% of those specifically focus on drug interactions. The EU mandates risk management plans for every approved drug. Japan followed suit in 2012. But here’s the catch: labeling hasn’t kept up. Most drug inserts still list interactions in dense paragraphs, buried under dozens of other warnings. There’s no standard format. No color coding. No priority ranking. That’s why patients miss them. New systems are being tested - like blockchain for reporting and genomic data to predict who’s at risk based on their DNA. The NIH’s Pharmacogenomics Research Network-2 is already analyzing 15,000+ cases to see how genes affect how people metabolize drugs. In the future, your doctor might check your genetic profile before prescribing - not just your current meds.What You Can Do Today
Don’t wait for a warning label to change. Take action now:- Make a full list of every medication, supplement, and OTC drug you take - including vitamins and herbal products.
- Bring that list to every appointment - even if you’ve seen the doctor before.
- Ask your pharmacist to run a drug interaction check every time you get a new prescription.
- Use free tools like GoodRx or Medscape’s interaction checker. They’re not perfect, but they catch 80% of major risks.
- If you experience unexplained muscle pain, dark urine, dizziness, or unusual bleeding after starting a new drug, contact your provider immediately. It could be an interaction.
Drugs save lives. But they also carry hidden risks - risks that only show up after they’re in your hands. The system isn’t broken. It’s just slow. And until it gets faster, you have to be your own safety net.
What are the most common post-market drug interactions?
The most common include statin-azole antifungal combinations (like simvastatin and fluconazole), statins with grapefruit juice, and blood thinners like apixaban with St. John’s Wort. These interactions often involve the CYP3A4 enzyme, which is responsible for breaking down about half of all prescription drugs. When this enzyme is blocked, drug levels spike, leading to toxicity.
Why don’t drug labels always warn about interactions?
Drug labels are written based on pre-market data, which is limited. Some interactions only appear after years of use, in older patients, or when combined with other drugs not tested together. Regulatory agencies update labels when new evidence emerges, but the process can take months or years. Meanwhile, many patients never read the full label - or the warning is buried in fine print.
Can I trust online interaction checkers like GoodRx?
Yes - but use them as a tool, not a final answer. Tools like GoodRx and Medscape are updated regularly and pull data from FDA reports and peer-reviewed studies. They’re highly accurate for major interactions. However, they may miss rare or newly emerging risks. Always confirm with your pharmacist or doctor, especially if you’re taking multiple medications.
How long does it take for a dangerous interaction to be discovered?
It varies. Some are caught within months - like the Exalgo alcohol interaction, discovered after 18 months. Others take years. Benfluorex caused harm for 30 years before being withdrawn. The delay depends on how many people are taking the drug, how rare the side effect is, and how well the reporting system works. On average, major safety events emerge 2 to 5 years after approval.
Are herbal supplements safer than prescription drugs when it comes to interactions?
No. Many people assume supplements are harmless, but that’s not true. St. John’s Wort, garlic, ginkgo, and echinacea can all interfere with prescription drugs. St. John’s Wort reduces the effectiveness of birth control, antidepressants, and blood thinners. In one case, a patient on Eliquis had life-threatening bleeding after taking it. Supplements aren’t tested for interactions like drugs are - so they’re riskier than most realize.
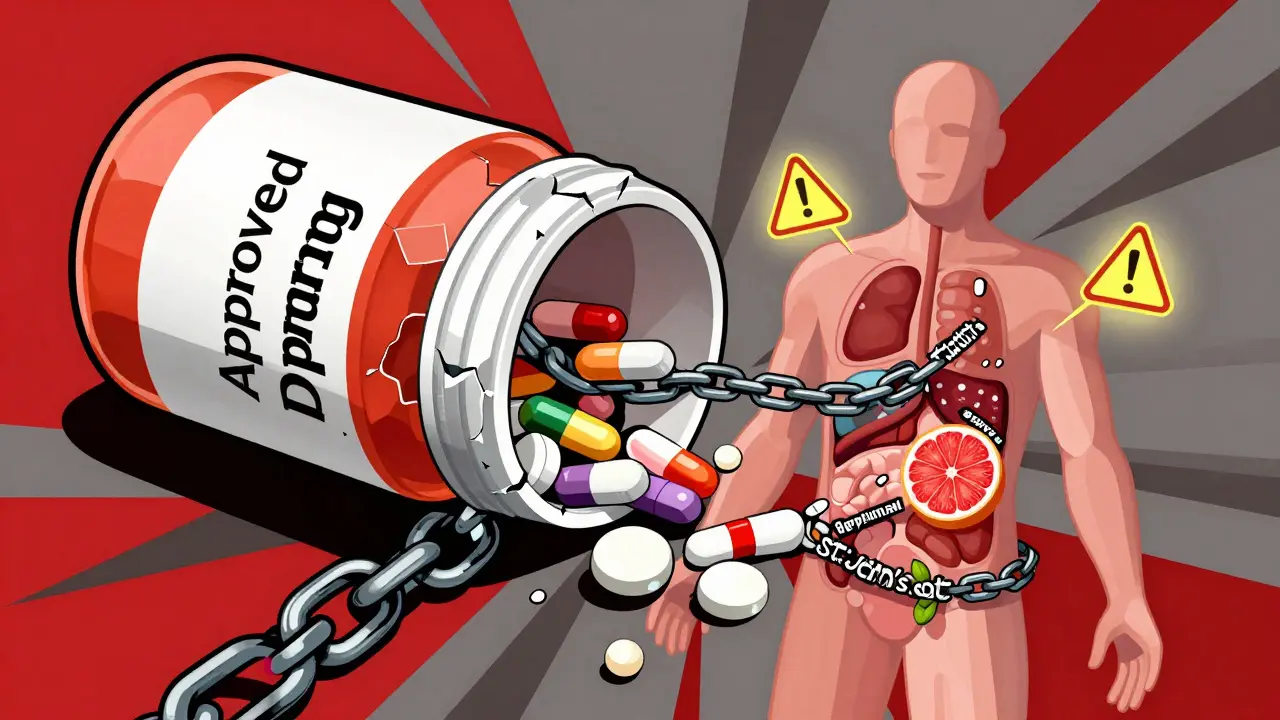




So let me get this straight - we’re trusting pharmaceutical companies to test drugs on a few thousand people and then unleash them on millions? And we’re supposed to be grateful when they finally admit they missed something? This isn’t oversight. It’s corporate negligence wrapped in a lab coat. The system isn’t slow - it’s designed to be blind until the body count rises. I’m not surprised. I’m just done pretending this is science.
March 2Megan Nayak